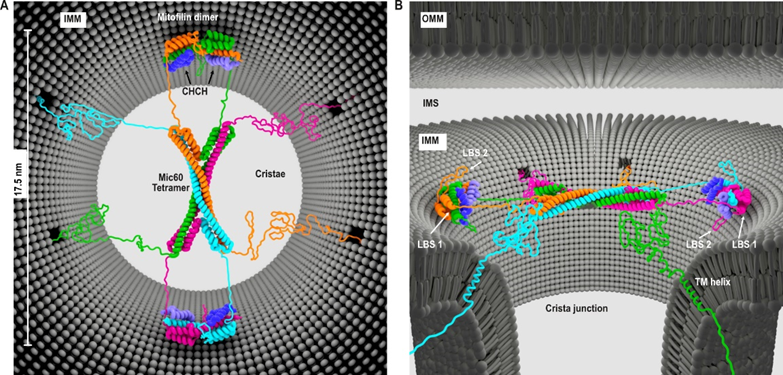
P06 - Structural and mechanistic studies on the machineries governing the shape of the inner mitochondrial membrane
The complex architecture of mitochondrial cristae is controlled by sophisticated protein machineries that remodel the inner mitochondrial membrane (IMM). Two prominent examples for such machineries are the dynamin-like Mgm1/OPA1 GTPase and the mitochondrial contact site and cristae organizing system (MICOS). In previous work, we determined the structure of a fungal Mgm1 homologue, clarified its assembly mode into a helical filament and suggested a model of how such filaments constrict cristae tubes in a GTPase-dependent fashion. In the last funding period, we solved structures of several parts of the MICOS component Mic60, structurally characterized its assembly with Mic19 and proposed a mechanism of how the Mic60-Mic19 subcomplex determines the architecture of mitochondrial crista junctions. Our experiments indicate that Mic60 exists in an auto-inhibited dimeric form, while Mic19 binding induces the transition to an active, tetrameric state.
In Aim 1 of this proposal, we will capitalize on our previous structural findings of the MICOS complex and apply a combination of biochemical and hydrogen deuterium exchange experiments to characterize the transition of Mic60 from a dimeric auto-inhibited to an active, Mic19-bound state. We will also study the impact of posttranslational modifications on the activation of Mic60. In Aim 2, we will structurally explore the auto-inhibited state of Mic60, employing data obtained from Aim 1. We will furthermore use single particle cryo electron microscopy to determine the structure of the Mic60-Mic19 tetramer. A comparison of the auto-inhibited and activated structures will reveal the molecular basis of Mic60 activation. In the final Aim 3 and in collaboration with groups of the consortium, we will translate the biochemical and structural findings of Aim 1 and 2 into a cellular understanding of Mic60 function. In particular, we aim to understand if and how the Mic60-Mic19 subcomplex regulates crista junction formation and disassembly. To this end, the impact of structure-based mutations in Mic60 and Mic19 on cristae junction architecture and dynamics will be studied in yeast and human cell lines. The proposed experiments will reveal new insights into the mechanism of crista junction formation, dynamics and turnover.