P08 - Influence of oligodendrocyte factors in vivo on the heterogeneity of the inner mitochondrial membrane in axons of retinal ganglion cells and white matter tracts
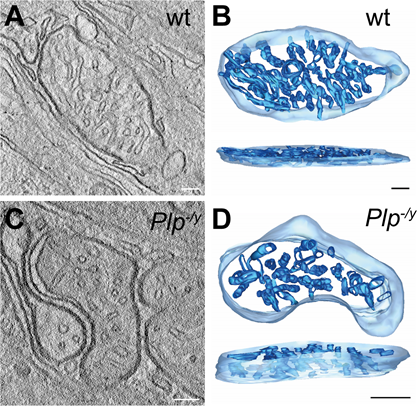
Saltatory action potential propagation and axonal transport in central nervous system axons depends on myelination by oligodendrocytes which support the axonal energy homeostasis by several means. Compromised myelin function causes neurodegeneration accompanied by axonal swellings and alterations in the morphology and function of axonal mitochondria. Retinal ganglion cells which extend their axons along the highly myelinated optic nerve appear to be especially vulnerable to mitochondrial dysfunction. We will continue our studies on the local mitochondrial heterogeneity in these neurons in a dysmyelinating mouse model lacking the proteolipid protein (PLP). Oligodendrocytes support the axonal energy metabolism by not only providing metabolites such as lactate and pyruvate, but also by the release of exosomes which contain proteins and nucleic acids. In the Plp-deficient mouse (Plp−/y) less exosomes are secreted which also show an altered proteome signature and fail to support axonal transport. One critical component lacking in exosomes of the Plp−/y mouse is the deacetylase Sirtuin 2 (SIRT2). This enzyme is highly expressed in oligodendrocytes. Exosome-mediated transfer of SIRT2 was recently shown to result in deacetylation of inner mitochondrial membrane proteins and enhance axonal ATP production. Therefore, we aim at investigating the role of protein acetylation in general and specifically the influence of SIRT2 on axonal mitochondria function in the Plp-deficient mouse. For this purpose, we plan to isolate neuronal mitochondria from white matter tracts to analyze their proteome and acetylome profile as well as their respiration in the Plp-deficient mouse. To address specifically the role of SIRT2 we plan to apply AAV-mediated gene transfer of SIRT2 into CNS neurons circumventing the Plp-deficient oligodendrocytes. To assess effects of SIRT2 delivery on axonal mitochondria we will localize recombinant SIRT2 in axons and investigate axonal mitochondrial structure by analysis of their shape, distribution and cristae morphology. In addition, we will quantify axonal swellings as indicators for impaired axonal transport. In summary, we hope to gain insight into critical changes of the axonal mitochondrial protein composition and the state of their posttranslational modification to better understand inner mitochondrial membrane heterogeneity as a consequence of impaired axo-glial support.