P07 - Role of the inner mitochondrial membrane in innate immune signaling paradigms
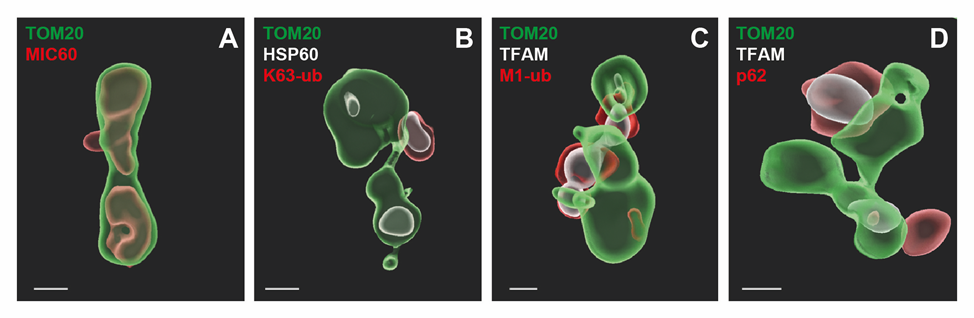
Mitochondria are essential for the maintenance of cellular and organismal integrity, based on their manifold functions in regulating cellular metabolism and coordinating cell fate decisions. More recently, mitochondria have been recognized to play a pivotal role in orchestrating innate immune signaling pathways. They are predestined for this task by virtue of their motility and dynamics, facilitating interorganellar communication. As organelles of proteobacterial origin, mitochondria harbor danger signals and need to be protected from immune surveillance. On the other hand, mitochondria have been co-opted by eukaryotic cells to react to cellular damage and to promote efficient immune responses. There are multiple conundrums concerning the mechanisms mitochondria evolved to affect cellular signaling. In several paradigms, a signaling platform is assembled at the outer mitochondrial membrane (OMM), involving posttranslational modifications of OMM proteins and recruitment of specific proteins. However, to adapt mitochondrial functions to context-specific cellular demands during signaling, information needs to be transmitted from the OMM to the inner mitochondrial membrane (IMM). How alterations at the OMM are signaled to the IMM and the mitochondrial matrix has largely remained unexplored.
We recently identified an innate immune signaling pathway that employs mitochondria for signal amplification and shuttling of activated transcription factors to the nucleus. Binding of the anti-apoptotic tumor necrosis factor (TNF) to its receptor at the plasma membrane induces recruitment of signaling components to the OMM and their activation by ubiquitination and phosphorylation. This remodeling of the OMM goes along with an increase in the mitochondrial membrane potential and respiratory activity and with the maintenance of cristae integrity under pro-apoptotic conditions, indicating signal transmission from the OMM to the IMM. Here, we aim at identifying components of the IMM implicated in anterograde (outside-in) mitochondrial signaling and to determine the underlying mechanisms (AIM 1). A promising target in this context is the mitochondrial contact site and cristae organizing system (MICOS), based on its role in forming contact sites between the IMM and OMM. In AIM 2 we will explore mitochondria as a source of danger signals, such as mtDNA, as an example for retrograde (inside-out) signaling and characterize determinants of IMM integrity that prevent the release of mtDNA in different stress paradigms. With these experiments combining state-of-the-art approaches from cell biology, advanced cellular imaging, biochemistry, proteomics and lipidomics, we expect to reveal key aspects of the IMM architecture and function in innate immune signaling.